Pyrrole-Modified Porphyrins
 For the first ~100 years of synthetic porphyrin chemistry, no porphyrinoids with a non-tetrapyrrolic chromophore framework were known. This has dramatically changed. In fact, heteroporphyrins, porphyrin isomers, expanded or contracted porphyrins, and pyrrole-modified porphyrins (PMPs), porphyrin analogues containing non-pyrrolic heterocylces, are leading topics in contemporary porphyrin research. This renaissance invigorated a seemingly mature research area because of the potentially dramatic influences the replacement of a pyrrole may have on the electronic structures of the resulting porphyrinoid. PMPs may also carry functionalities at their periphery that could be utilized for sensing or bioconjugation.
For the first ~100 years of synthetic porphyrin chemistry, no porphyrinoids with a non-tetrapyrrolic chromophore framework were known. This has dramatically changed. In fact, heteroporphyrins, porphyrin isomers, expanded or contracted porphyrins, and pyrrole-modified porphyrins (PMPs), porphyrin analogues containing non-pyrrolic heterocylces, are leading topics in contemporary porphyrin research. This renaissance invigorated a seemingly mature research area because of the potentially dramatic influences the replacement of a pyrrole may have on the electronic structures of the resulting porphyrinoid. PMPs may also carry functionalities at their periphery that could be utilized for sensing or bioconjugation.
The total synthesis of PMPs is possible. However, we focus our attention to the development of an alternative strategy, the step-by-step derivatization of readily available synthetic porphyrins, such as meso-tetraaryl- and β-alkylporphyrins. We paraphrased this approach as ‘the breaking and mending of porphyrins’.
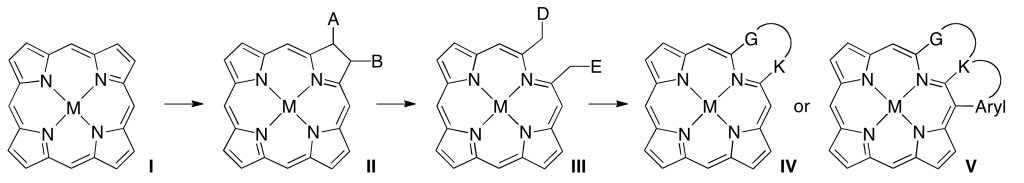
Addition of a reagent to the β,β’-double bond of a porphyrin (I) converts it to a functionalized chlorin (II). The functional group provides handles for subsequent β,β’-ring cleavage reactions (III). Either in situ or in a separate step, the functionalities thus generated are converted to form a PMP (IV). Thus, one pyrrolic unit was formally replaced by a non-pyrrolic heterocycle that may be ring-contracted or expanded relative to the pyrrole in I, or not a ring at all. Product IV may lend itself to further derivatization, such as the formation of β-to-aryl linkages (V). A second pyrrolic moiety may be susceptible to an analogous conversion. This step-wise approach is controlled, flexible, and allowed for the synthesis of singly and doubly pyrrole-modified chromophores containing a wide range of heterocycles.
An overview over the ‘breaking and mending’ strategy is also available: Brückner, C. ‘The Breaking and Mending of meso-Tetraarylporphyrins: Transmuting the Pyrrolic Building Blocks’ Acc. Chem. Res. 2016, 49, 1080−1092.
Some of the PMP classes presented by us are shown below.

A comprehensive review of PMPs is available: Brückner, C.; Akhibe, J.; Samankumara, L. ‘Syntheses of Porphyrin Analogs Containing Non-Pyrrolic Heterocycles’, in Handbook of Porphyrin Science; Eds. Kadish K. M.; Smith, K. M.; Guilard, R.; World Scientific: River Edge, NY, 2014; Vol. 31, 1–276.
See publication page for a listing of all publications.
Current Projects-Synthesis
Current synthetic projects involve the search for novel ways to activate a porphyrin (using, for instance, ozone, or organometallic oxidants) and the search for the novel ways to convert established derivatives into new classes of PMPs. Next to our workhorse meso-arylporphyrins, we have also begun to study ß-octaethylporphyrin as starting material toward pyrrole-modified porphyrin. Also most pressing is our current search for the generalized solubilization of the porphyrins for their application in biological settings.